 Methoxide
anion
Methoxide
anion
Basis Set Effect
操作low-level 結構最佳化時會使用medium-sized basis set (ex: HF or B3LYP with 6-31G(d))
在最佳化結構下執行的frequency job,必須使用同一個method和basis set (Opt Freq in Gaussian)
執行very high level單點能量計算時 (ex: MP4 or QCISD(T)),使用large basis set
Minimal Basis Sets: use fixed-size atomic-type orbitals
ex: STO-3G
Split Valence Basis Sets: have two sizes of basis function for each valence orbital
ex: 6-31G、6-311G
Polarized Basis Sets: add orbitals with angular momentum to the description of each atom
ex: 6- 31G(d)、6-31G(d,p)
Diffuse Functions: allow orbitals to occupy a larger region of space
ex: 6-31G+(d)、6-31G++(d)
High Angular Momentum Basis Sets: add multiple polarization funtions per atom to the triple zeta basis set
ex: 6-311++G(3df,3pd)、6-311++G(3df,2df,p)
Example 5.1: Methanol vs. Methoxide Anion Optimization (HF method)
Methanol Methoxide
anion
Methoxide
anion
Methanol 6-31G(d) 6-31+G(d) Experiment
CO bond 1.3996 1.4015 1.427±0.007
(1 3) CH bond 1.0811 1.0804 1.096±0.01
OH bond 0.9463 0.9463 0.956±0.015
COH angle 109.406 110.4137 108.9±2.0
(4 1 5) HCH angle 108.7121 109.0762 109.3±0.75
(2 1 3) OCH angle 107.159 106.9084
Diffuse functions 對結構最佳化的影響很小
Methoxide anion 6-31G(d) 6-34+G(d) 6-311++G(3df,2pd)
CO bond 1.3105 1.3304 1.3223
CH bond 1.133 1.121 1.1209
HCH angle 101.5572 103.4298 103.2904
OCH angle 116.5485 114.9919 115.1097
Example 5.2: PO Bond Distance (B3LYP method)
Basis Set Bond Length (Angstroms)
6-31G(d) 1.4986
6-311G(d) 1.4915
6-311G(2d) 1.4819
6-311G(2df) 1.4796
6-311G(3df) 1.4759
Experiment 1.476
如要精確計算出此結構,則必須用high angular momentum basis sets
Exercise 5.1: HF Bond Length (mp4 method)
Basis Set Bond Length
6-31G(d) 0.935
6-31G(d,p) 0.9211
6-31+G(d,p) 0.9264
6-31++G(d,p) 0.9264
6-311G(d,p) 0.9132
6-311++G(d,p) 0.9172
6-311G(3df,3pd) 0.9137
6-311++G(3df,3pd) 0.9174
Experiment 0.917
6-311G的計算結果比6-31G精確
加上2個diffuse function效果更好
polarization對計算結果沒有明顯的影響
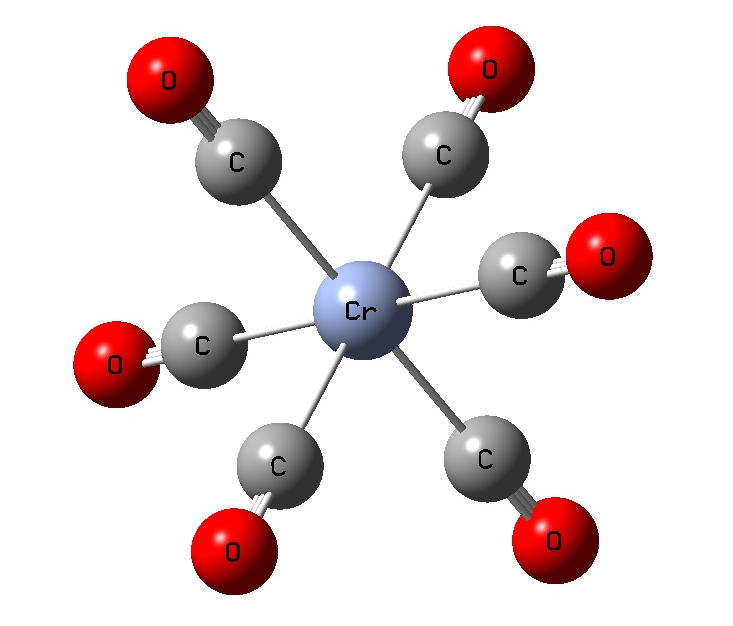
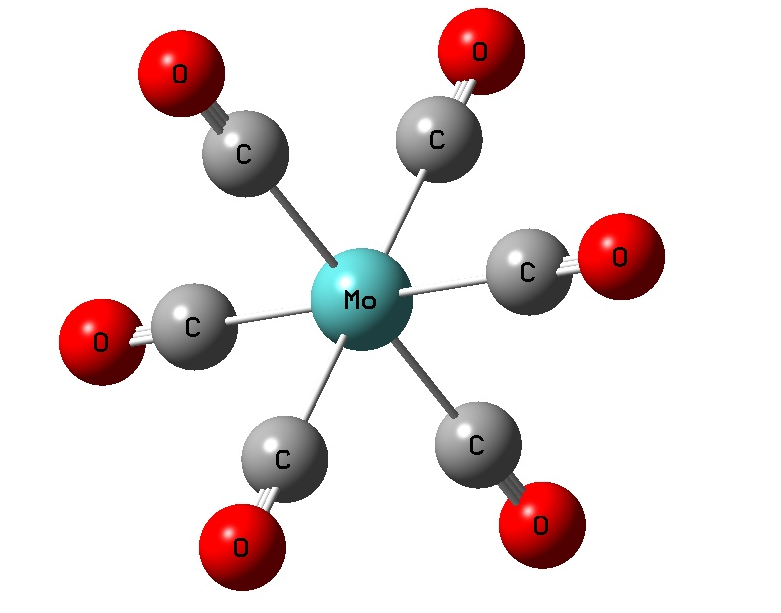
Exercise 5.2: Periodic Trends in Transition Metal Complexes (M(CO)6) (HF/LANL2DZ)
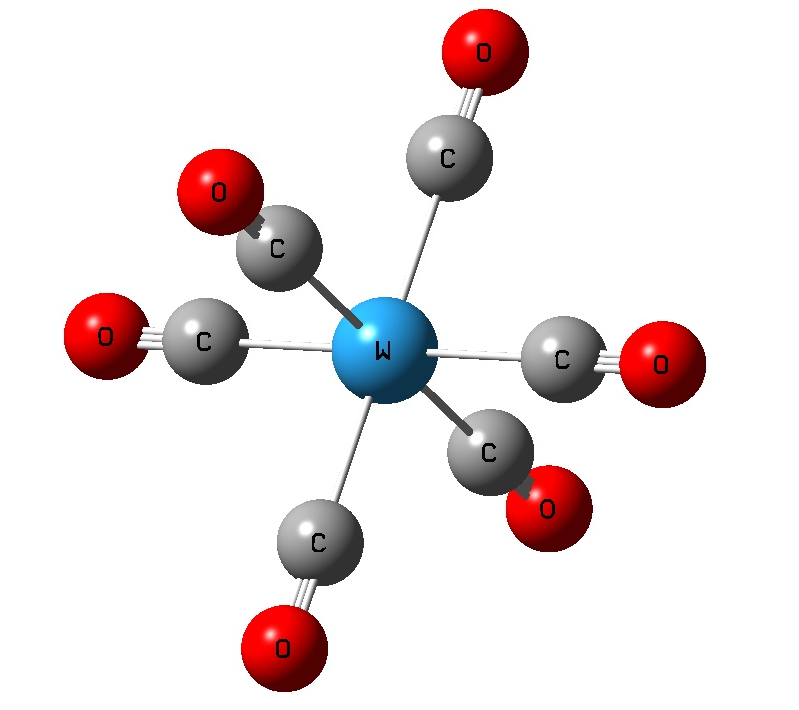
M M-C bond C-O bond M-C-O C-M-C
Cr 1.9789 1.1411 180.0 90.0, 180.0
Mo 2.1047 1.1428 180.0 90.0, 180.0
W 2.0774 1.1445 180.0 90.0, 180.0
Exercise 5.3: Basis Set Effect on NMR Calculations (Benzene)
以B3LYP/6-31G(d)做結構最佳化,使用HF方法搭配各種basis set計算NMR,比較結果的不同
Absolute Shielding Value
Model Benzene TMS Relative Shift
HF/6-31G(d) 72.6433 195.1196 122.4763
HF/6-31G(d,p) 72.9133 196.6246 123.7113
HF/6-31+G(d,p) 72.7219 197.2420 124.5201
HF/6-31++G(d,p) 72.7437 197.1381 124.3944
HF/6-31+G(2d,p) 57.6198 188.7880 131.1682
Observed chemical shift 130.9
6-311+G(2d,p)最準,而且準很多!!
Exercise 5.4: Geometry of N,N-Dimethylformamide (HF method)
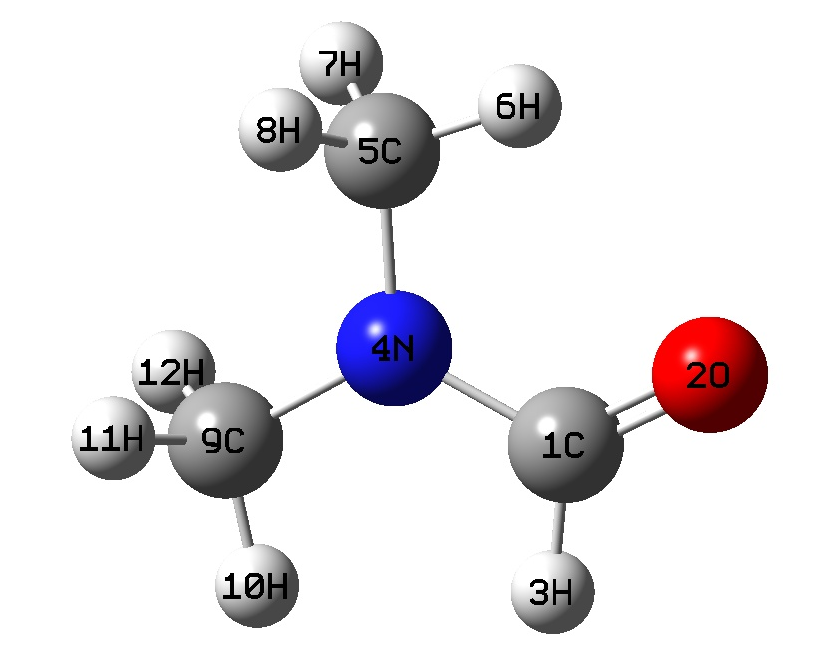 N,N-Dimethylformamide
(not real structure)
N,N-Dimethylformamide
(not real structure)
Parameter STO-3G 6-31G(d) 6-31++G(d,p) Experiment
R(O-C1) 1.218 1.1965 1.2001 1.224±.003
R(C1-N) 1.4097 1.3486 1.3463 1.391±.006
R(C5-N) 1.4643 1.4462 1.4478 1.453±.004
R(C9-N) 1.4631 1.4424 1.4441 1.453±.004
A(N-C1-H3) 111.3903 112.5242 112.7897 117.0±2.8
A(O-C1-N) 124.3456 125.9352 125.9022 123.5±0.6
A(O-C1-H3) 124.2641 121.5406 121.3081 119.5±3.4
A(C1-N-C5) 119.8977 120.6005 120.8288 120.8±0.3
A(C1-N-C9) 121.7929 121.9703 121.8041 122.3±0.4
A(C5-N-C9) 118.3091 117.4291 117.367 113.9±0.5
ΣA(C-N-C) 359.9997 359.9999 359.9999 357.0±1.2
D(C9-N-C1-H3) 0.1749 0.0926 0.0856 11±4
D(C5-N-C1-O) -0.0181 -0.075 -0.0608 16±5
Imaginary Frequencies 1 0 0
Is Ground State Planar NO YES YES NO